0814 islets of langerhans and diabetes mellitus type 1 medical images for powerpoint
Encourage great feats with our 0814 Islets Of Langerhans And Diabetes Mellitus Type 1 Medical Images For Powerpoint. Embolden them to act fearlessly.
- Google Slides is a new FREE Presentation software from Google.
- All our content is 100% compatible with Google Slides.
- Just download our designs, and upload them to Google Slides and they will work automatically.
- Amaze your audience with SlideTeam and Google Slides.
-
Want Changes to This PPT Slide? Check out our Presentation Design Services
- WideScreen Aspect ratio is becoming a very popular format. When you download this product, the downloaded ZIP will contain this product in both standard and widescreen format.
-

- Some older products that we have may only be in standard format, but they can easily be converted to widescreen.
- To do this, please open the SlideTeam product in Powerpoint, and go to
- Design ( On the top bar) -> Page Setup -> and select "On-screen Show (16:9)” in the drop down for "Slides Sized for".
- The slide or theme will change to widescreen, and all graphics will adjust automatically. You can similarly convert our content to any other desired screen aspect ratio.
Compatible With Google Slides

Get This In WideScreen
You must be logged in to download this presentation.
PowerPoint presentation slides
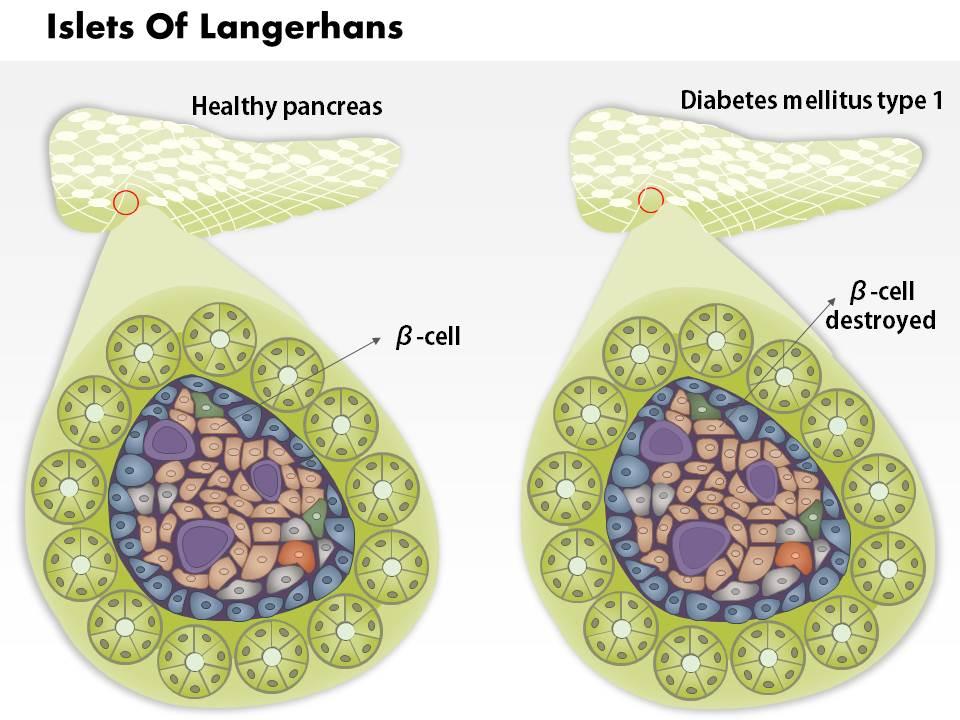
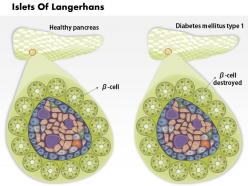
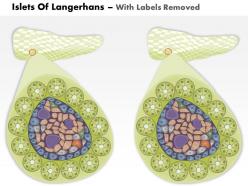
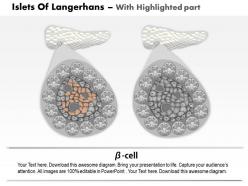
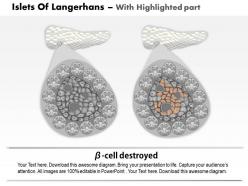
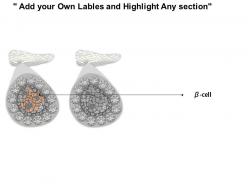
We are proud to present our 0814 islets of langerhans and diabetes mellitus type 1 medical images for powerpoint. This medical image has been developed to show the Islets Of Langerhans And Diabetes Mellitus Type 1. To display this disease, we have used graphic of healthy pancreas and one affected with type-1 diabetes. This image also displays the condition of ? cells in both healthy and diabetic conditions. Use this image to display this type 1 diabetes condition in any medical presentation.
People who downloaded this PowerPoint presentation also viewed the following :
0814 islets of langerhans and diabetes mellitus type 1 medical images for powerpoint with all 9 slides:
Get a convenient format with our 0814 Islets Of Langerhans And Diabetes Mellitus Type 1 Medical Images For Powerpoint. They are designed to be functional.
-
Use of different colors is good. It's simple and attractive.
-
Design layout is very impressive.