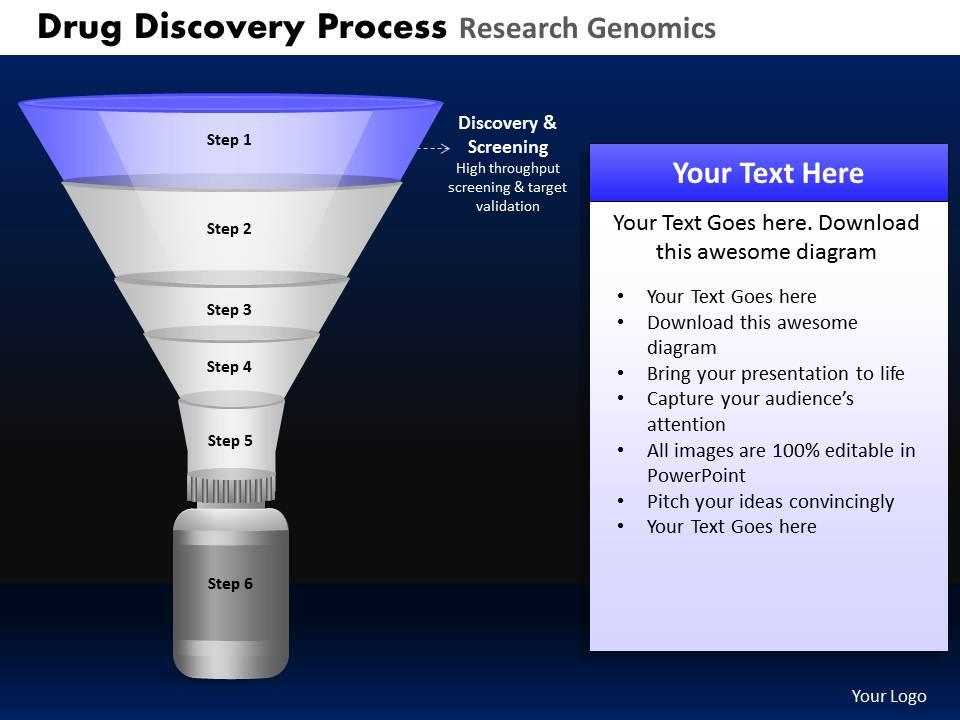
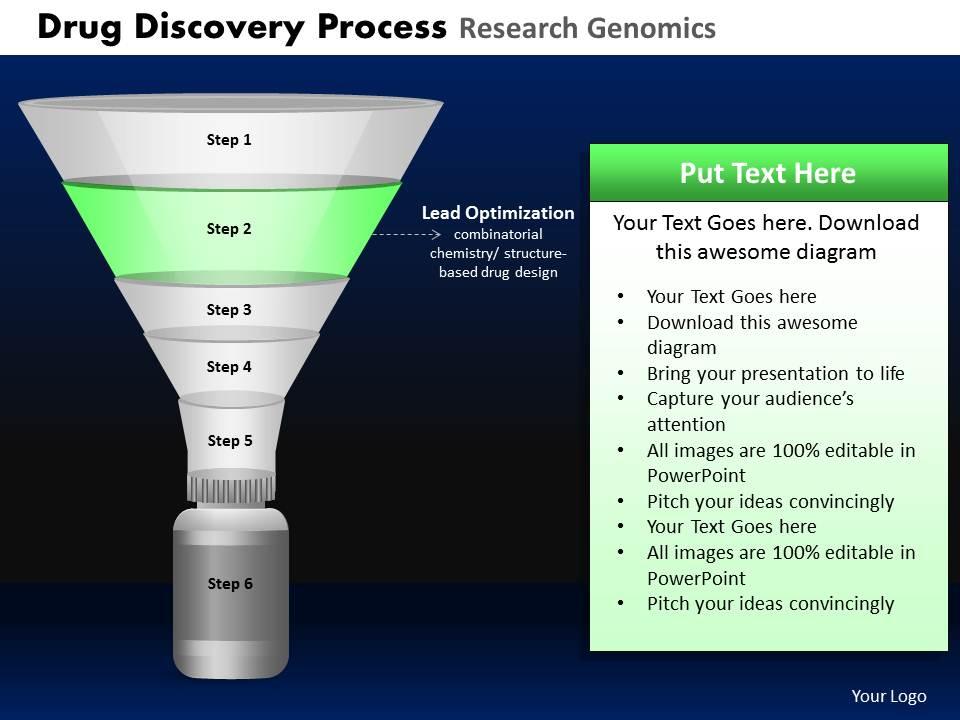
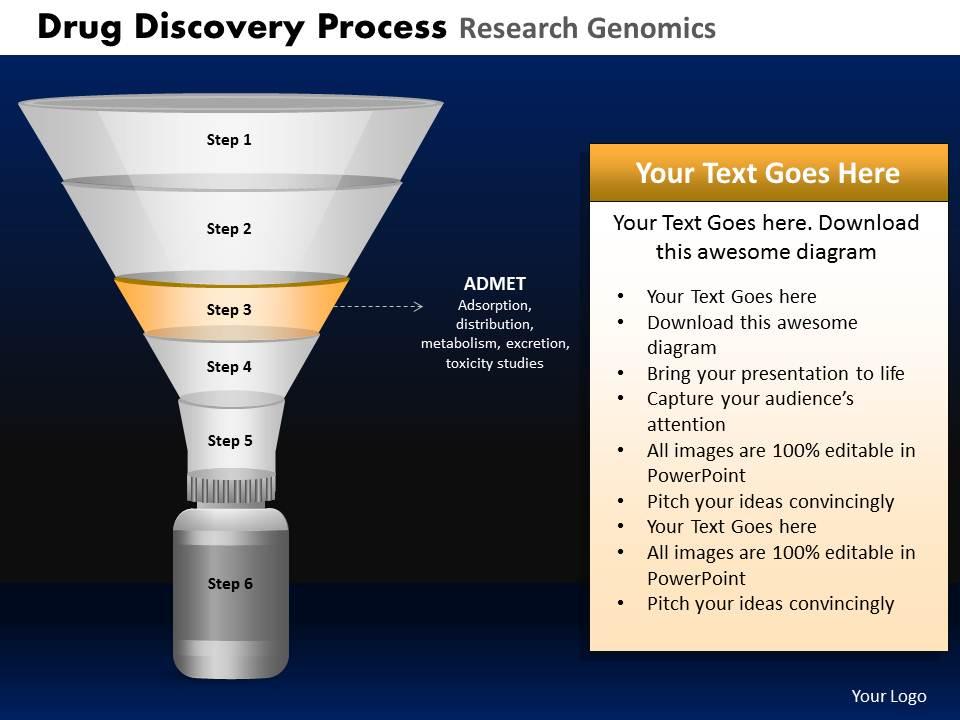
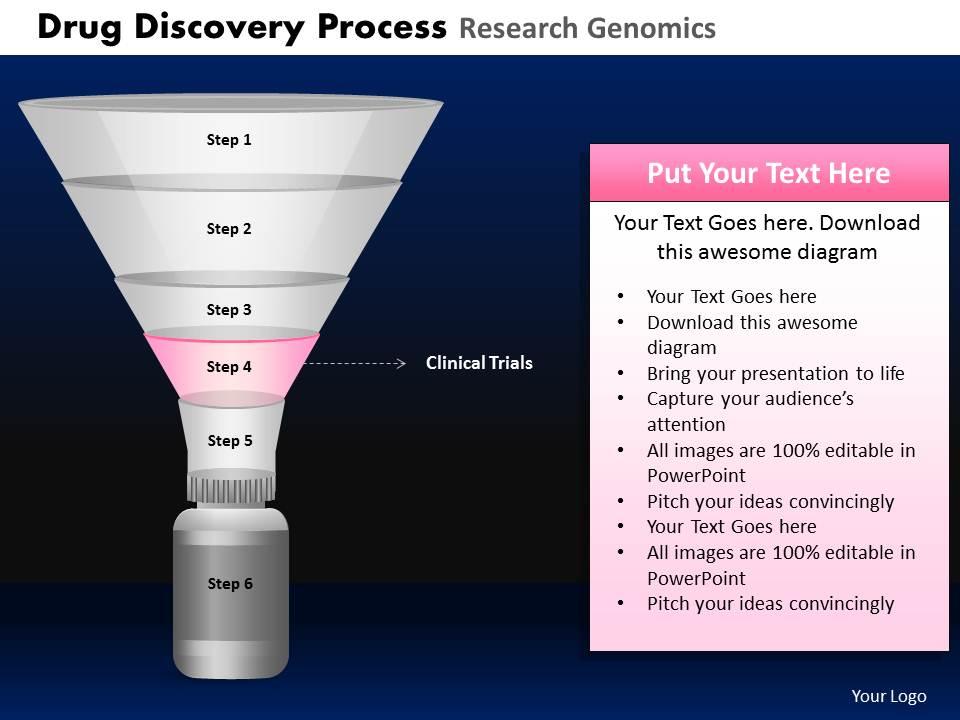
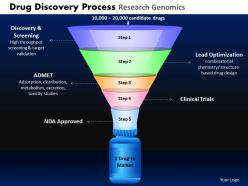
Drug discovery process research genomics powerpoint slides and ppt templates db
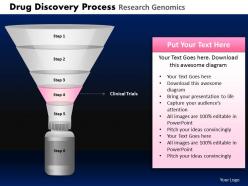
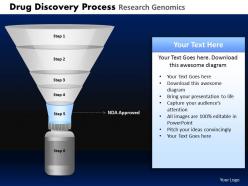
Give your ideas some tender loving care. Caress them with our Drug Discovery Process Research Genomics Powerpoint Slides And Ppt Templates DB.
You must be logged in to download this presentation.
 Impress your
Impress your audience
Editable
of Time
PowerPoint presentation slides
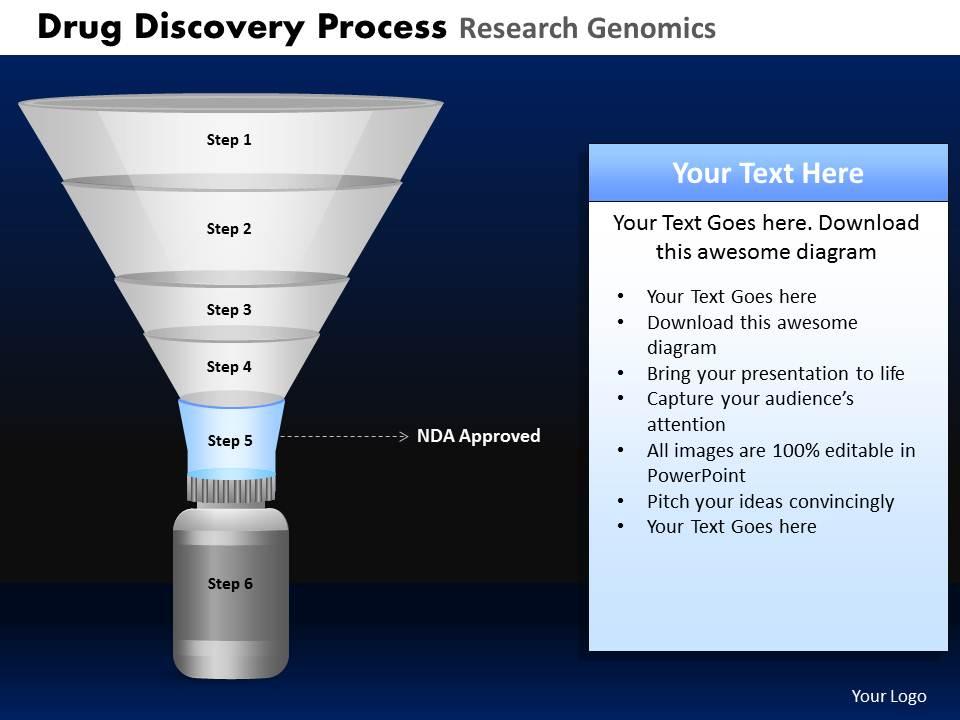
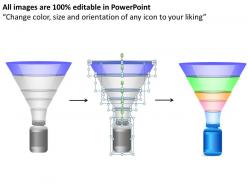
These high quality, editable pre-designed Drug Discovery Process Research Genomics Powerpoint Slides And Ppt Templates DB powerpoint slides and powerpoint templates have been carefully created by our professional team to help you impress your audience. Each graphic in every powerpoint slide is vector based and is 100% editable in powerpoint.Each and every property of any slide - color, size, shading etc can be modified to build an effective powerpoint presentation. Use these slides to convey complex business concepts in a simplified manner. Any text can be entered at any point in the powerpoint slide. Simply DOWNLOAD, TYPE and PRESENT
People who downloaded this PowerPoint presentation also viewed the following :
Drug discovery process research genomics powerpoint slides and ppt templates db with all 10 slides:
Crown your thoughts with our Drug Discovery Process Research Genomics Powerpoint Slides And Ppt Templates DB. They will rule the minds of your team.
-
Really like the color and design of the presentation.
-
Helpful product design for delivering presentation.